UPDATE August 22, 2023: The U.S. Preventive Services Task Force (USPSTF) issued its final statement for PrEP to prevent HIV, giving it the highest recommendation, an A grade. Of note, the updated recommendation includes long-acting injectable PrEP, or pre-exposure prophylaxis.
The USPSTF statement reads in part:
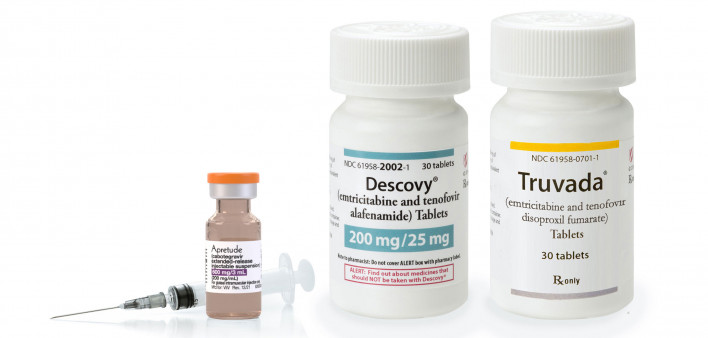
Based on its review of the evidence, the Task Force recommends that health care professionals prescribe PrEP to people at increased risk for HIV to help prevent HIV. This is an A grade.… PrEP is a safe, highly effective way to help prevent HIV in people who do not have HIV but are at increased risk of getting it. There are now three FDA-approved PrEP medications in the United States—two are pills, and one is a shot.…
“We’re still seeing that many people who could benefit from PrEP aren’t receiving this highly effective medication, particularly in Black, Hispanic and Latino communities,” says Task Force member James Stevermer, MD, MSPH. “It’s critical for health care professionals to keep these disparities in mind and to have conversations with patients who are at risk about proven ways to prevent HIV, including taking PrEP.”
The Affordable Care Act (ACA, or Obamacare) requires health insurance plans to cover preventive health care services that have received an A or B grade from the USPSTF. However, a Texas court case seeks to end coverage of some services.
Click here to read the final recommendation, evidence review and summary for patients in JAMA, which were accompanied editorials about long-acting PrEP, its potential obstacles and the benefits of more prevention choices.
At the end of last year, USPSTF issued a draft recommendation regarding injecting PrEP. Below is that original article.
DECEMBER 13, 2022: Long-acting injectable PrEP to prevent HIV received an A grade in an updated draft recommendation issued today by the U.S. Preventive Services Task Force (USPSTF), an independent panel of health care experts.
The grade is important because the Affordable Care Act (ACA, or Obamacare) requires that private insurers and ACA-approved health plans cover preventive services that receive an A or B grade from the USPSTF. This means that injectable PrEP (pre-exposure prophylaxis) will be available to more people and without cost-sharing.
The task force gave oral PrEP (taken as daily tablets) an A grade in 2019. The update would add another HIV prevention option for folks who prefer not to take the tablets. Increasing the options and availability will also help address health inequities, noted the USPSTF.
“Many people who would benefit from PrEP are not receiving this highly effective medication. This is especially true of Black, Hispanic and Latino communities,” said USPSTF member Martha Kubik, PhD, RN, in the recommendation. “These inequities must be addressed to achieve the full benefit of PrEP.”
The Food and Drug Administration approved Apretude (cabotegravir) as the first long-acting injectable PrEP in December 2021. Administered every other month (six times a year), Apretude is manufactured by ViiV Healthcare.
The draft recommendation released today is open for public comment through January 17, after which the USPSTF will issue a final recommendation.
“Health care professionals can help protect their patients at increased risk for HIV by prescribing PrEP,” said task force member John B. Wong, MD, in the draft recommendation. “The availability of multiple effective PrEP medications is great news for patients and a positive step to helping reduce the impact of HIV in the U.S.”
HIV advocates praised the updated recommendation but cautioned that government action was needed to ensure access to the injectable option. As activist group PrEP4All wrote in a statement:
“The biggest issue with making [long-acting cabotegravir, CAB-LA] more widely available has everything to do with how much it costs. At approximately $22,500 USD for 6 shots annually, public health departments with PrEP access programs for the un- and under-insured will not be able to add it as an option for most people who want to use it. Furthermore with generic [tenofovir disoproxil fumarate/emtricitabine, TDF/FTC, or oral PrEP] now less than $30 for a 30-day supply, we are certain to see insurance companies continue to put up barriers to access, like prior authorization without a medical reason for needing CAB-LA over TDF/FTC. These FDA-approved generic TDF/FTC medications are excellent options for many potential PrEP users, but we do not want to repeat the same unnecessary delays in scale-up for CAB-LA that we have seen for daily oral formulations. We have yet to even fulfill the promise of daily oral PrEP; in 2020 only 25% of the 1.2 million Americans most in need of PrEP had received a prescription. Among potential Black and Latinx PrEP users, only 9% and 16% had a prescription, respectively.
“But there are other issues with the insurance market to cover PrEP meds and services. Despite the 2019 USPSTF ruling that gave an A grade to daily oral PrEP and the 2021 FAQ issued by Center for Medicare and Medicaid Services clarifying for payers that covering PrEP meant medications, visits and labs, PrEP4All to date has documented over 100 cases of people still being charged for labs, and other organizations nationwide have identified more individuals. This year the Senate got involved, led by Senator Merkley (D-OR) and called on AHIP to answer for its member insurers who continue to charge PrEP users, effectively breaking the law. With an injectable that is so much more expensive than generic daily oral PrEP, we are likely to see this continue.
“So while PrEP4ALL welcomes this ruling, there is still no law or institution by which insurance companies can be held accountable to ensure those who want to take PrEP, in whatever form, will have access to it. The high price will also continue to make CAB-LA too expensive for public programs for un- and under-insured individuals. We have been working with many allies to hopefully have Congress fund a National PrEP Program, which would greatly expand access to even more people who want PrEP but are presently un- and/or under- insured, but until ViiV– the sole manufacturer of CAB-LA– and the US government negotiate a reasonable public health price, the intervention will likely remain out of reach through such an initiative.”
Other HIV advocacy groups echoed PrEP4All’s statement.
“We are extremely pleased that the USPSTF, at our request, has updated its recommendation for PrEP to prevent HIV and include newly approved long-acting drugs,” said Carl Schmid, executive director of the HIV+Hepatitis Policy Institute, in statement. “Due to the importance of preventing HIV and the superiority of long-acting PrEP compared to daily oral PrEP, the USPSTF acted in record speed. Now we urge insurers to ensure its coverage and offer it without cost-sharing. We are hearing many complaints by people who are still being charged cost-sharing for oral PrEP and its associated services. People seeking PrEP are facing too many barriers to access and we need state and federal regulators to take the necessary steps to correct these problems.”
To read about the USPSTF’s 2019 decision to give oral PrEP an A grade, see “Great News! Expert Panel Recommends PrEP for HIV Prevention.” And for a collection of related articles, click #PrEP, where you’ll find headlines including:
“Judge Rules That the Mandate to Cover PrEP for HIV Prevention Violates Religious Freedom,”
“Could Cabenuva and Apretude Injections Be Self-Administered?,”
“Leadership Changes at PrEP4All, the HIV Prevention Advocacy Group” and
“Most Women Know About PrEP, but Few Are Using It.”






1 Comment
1 Comment